[ad_1]
Disclaimer: Early release articles are not considered as final versions. Any changes will be reflected in the online version in the month the article is officially released.
Author affiliation: Virginia Polytechnic Institute and State University, Blacksburg, Virginia, USA (D.A. Conley, G. Blanvillain, K.E. Langwig, J.R. Hoyt); University of Wisconsin–Madison, School of Veterinary Medicine, Madison, Wisconsin, USA (J.L. Miller); Virginia Department of Wildlife Resources, Henrico, Virginia, USA (J.D. Kleopfer); US Geological Survey, National Wildlife Health Center, Madison, Wisconsin, USA (J.M. Lorch)
Detecting and assessing declines in elusive or rare species can be difficult. Early identification of populations in decline can help accelerate intervention strategies and reduce the likelihood of genetic bottlenecks, population extirpation, and trophic disturbances of ecologically important species (1). Snake fungal disease (SFD) is caused by the fungal pathogen Ophidiomyces ophidiicola and affects a broad range of snake species (2), causing skin lesions as the fungus invades tissues, sometimes leading to impaired movement, anorexia, and even death (3). Researchers have documented population impacts from SFD in 2 snake species (4,5), but the extent of mortality across snake species is likely underestimated due to the cryptic nature of snakes. We describe a multiyear mortality event associated with SFD in a rare species, the rainbow snake (Farancia erytrogramma), in Virginia and North Carolina, USA.
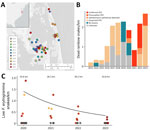
In spring 2019, regional biologists from the Back Bay region of North Carolina and Virginia reported 6 deceased F. erytrogramma snakes. In spring of 2020, we found an additional 6 snakes in the same area (Figure 1, panel A). After those events, we gathered additional records of dead F. erytrogramma snakes (Appendix Table 1) from the region and began regular surveys and sampling of F. erytrogramma snakes and other species for O. ophidiicola in 2020–2023 (Appendix).
We captured and swabbed snakes in accordance with previously published protocols (6) and used sterile procedures for disinfecting equipment (Appendix). We extracted DNA from and tested samples using quantitative PCR (qPCR) to determine the presence of O. ophidiicola, according to established methods (6,7). To quantify lesion severity for all live-captured snakes, we used an approach integrating snake size, lesion size, and lesion progression (Appendix Table 2, Figure 1).
All dead F. erytrogramma snakes screened by qPCR (n = 9) were positive for O. ophidiicola and had skin lesions characteristic of SFD. Necropsies on a subsample (n = 6) indicated all snakes examined had lesions consistent with O. ophidiicola infection, including thickening of the epidermis by eosinophilic necrotic cellular debris containing fungal hyphae. Snakes also had invasion by hyphae in deeper tissue, including the dermis (6/6) (Appendix Figure 2, panel A), underlying skeletal muscle (3/6), and in oral and nasal epithelium and tooth pulp (1/6). We also observed hyphae or fibrin thrombi within blood vessels in the dermis (5/6). Most snakes (4/6) were in good body condition, had large amounts of fat, and showed no signs of other serious pathologic processes. We considered SFD as the ultimate cause of death in all 6 snakes.
We also examined the number of live F. erytrogramma snakes captured over time using sampling data from standardized surveys conducted in 2020–2023 to assess preliminary trends while accounting for effort (Figure 1, panel C). We found a general decrease in the probability of detecting live F. erytrogramma snakes over time (log-scale year coefficient = −0.482 ± 0.226; p = 0.033) (Figure 1, panel C; Appendix Tables 3, 4).
A broader comparison of O. ophidiicola prevalence and severity of infection among other snakes in the community revealed that F. erytrogramma snakes were among the most infected species (Figure 2): O. ophidiicola prevalence was 80.1% 95% CIs 62.5%–92.5%, and log10 lesion severity was −1.71 (95%CI −1.94 to −1.50) (Appendix). Several other snake species also had notably high prevalence of O. ophidiicola and did not differ greatly from F. erytrogramma snakes (Figure 2, panel A) but were not found dead as part of the ongoing mortality event. Although live F. erytrogramma snakes were rare within the broader snake community (total live captures 16.4% [95% CI 11.3%–23%]; n = 25/153), they were disproportionately represented among dead snakes (87.5% [95% CI 52.9%–97.8%]; n = 7/8) found during the same period (2020–2023).
Observing wildlife mortality without obvious cause is rare and can indicate a more serious problem (8). We documented mortality of F. erytrogramma snakes using photographic, molecular, and histologic evidence, providing support that infection with O. ophidiicola is likely responsible. The F. erytrogramma snakes is considered a species of conservation concern, and although mortality in the species appeared to be ongoing as of 2023, the full extent of population declines remains uncertain.
O. ophidiicola was likely introduced to the United States in the early 1900s, although new (and possibly more virulent) strains have emerged recently (9). Increases in F. erytrogramma snake mortality could be the result of the introduction of more virulent strains of O. ophidiicola or shifts in environmental conditions since 2014 (10), but it is unclear why F. erytrogramma snakes appear particularly susceptible to infection (Figure 2). Nonetheless, the observed epizootic from a pathogen that has existed in North America for decades suggests SFD remains a threat to snake populations, which are a critical ecologic component of many ecosystems. Further research on the potential effects of O. ophidiicola would help clarify the impacts and trends of this disease on snake populations. Our study highlights the potential impact of disease-causing fungi such as O. ophidiicola on unmonitored, cryptic snake species like the rainbow snake.
Mr. Conley is a PhD student in the Department of Biological Sciences at Virginia Polytechnic Institute and State University. His primary research interests include herpetology, ecology, and infectious disease in wildlife.
[ad_2]
Source link